Tokyo, Japan, April 30, 2026. Craif Inc., a bio-AI startup focused on early cancer detection, today announced results from a multicenter study demonstrating that AI-analyzed urinary microRNA can detect early-stage lung cancer(LC) with high accuracy.
The study was conducted in collaboration with Associate Professor Yu Fujita of The Jikei University School of Medicine and Dr. Takashi Nojiri of Higashiosaka City Medical Center. The findings also show that preoperative urine samples can predict postoperative recurrence risk, and that postoperative changes in urinary microRNA levels may support recurrence monitoring. Results were published in npj Precision Oncology on April 18, 2026.
- Lung cancer remains difficult to detect at an early stage: Lung cancer is the leading cause of cancer-related mortality worldwide. Because early-stage disease is often asymptomatic, most patients are diagnosed after the disease has advanced.
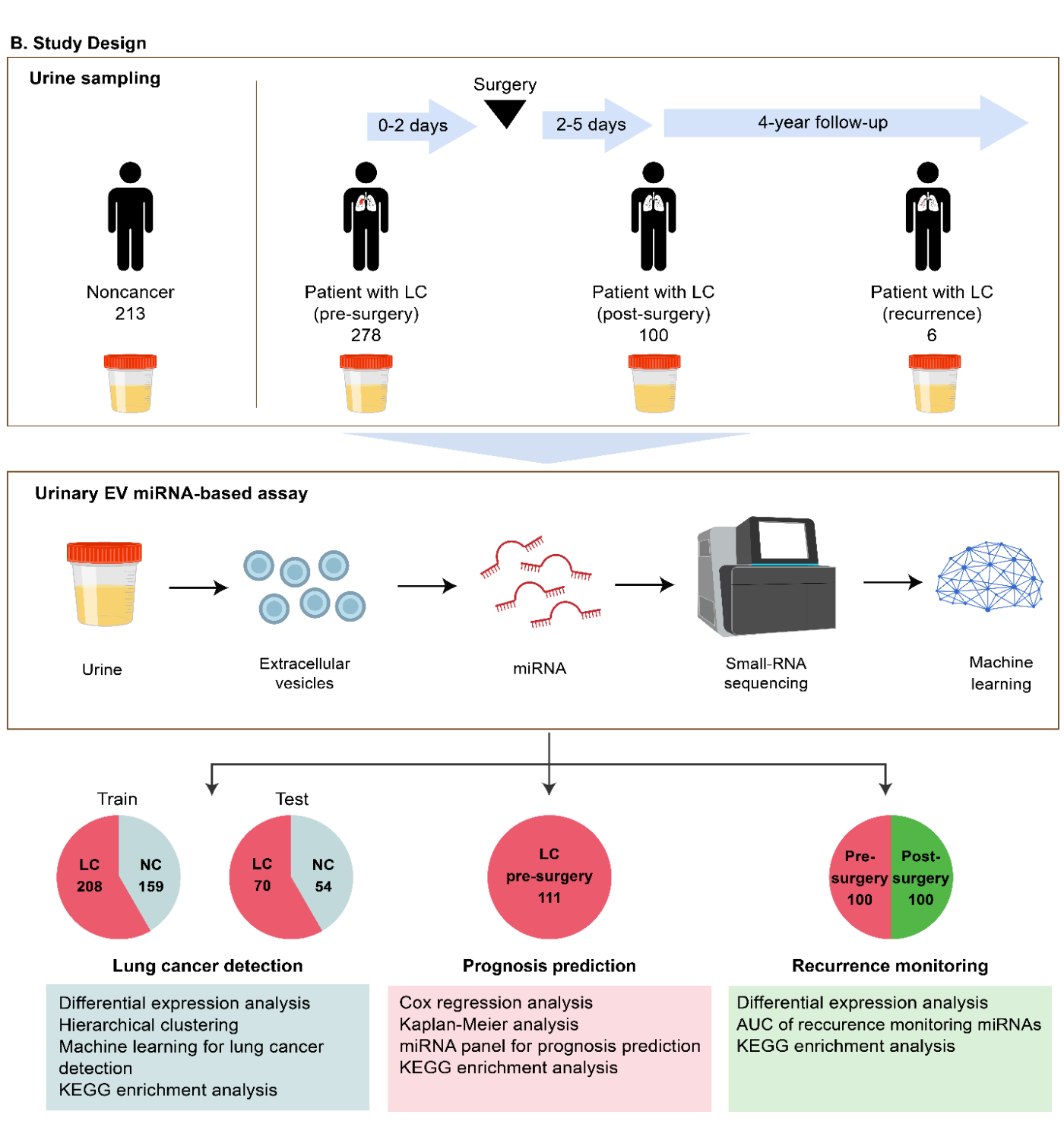
- Multicenter collaborative study: Urine samples from 278 with lung cancer patients (approximately half with early-stage disease) and 213 non-cancer controls were analyzed.
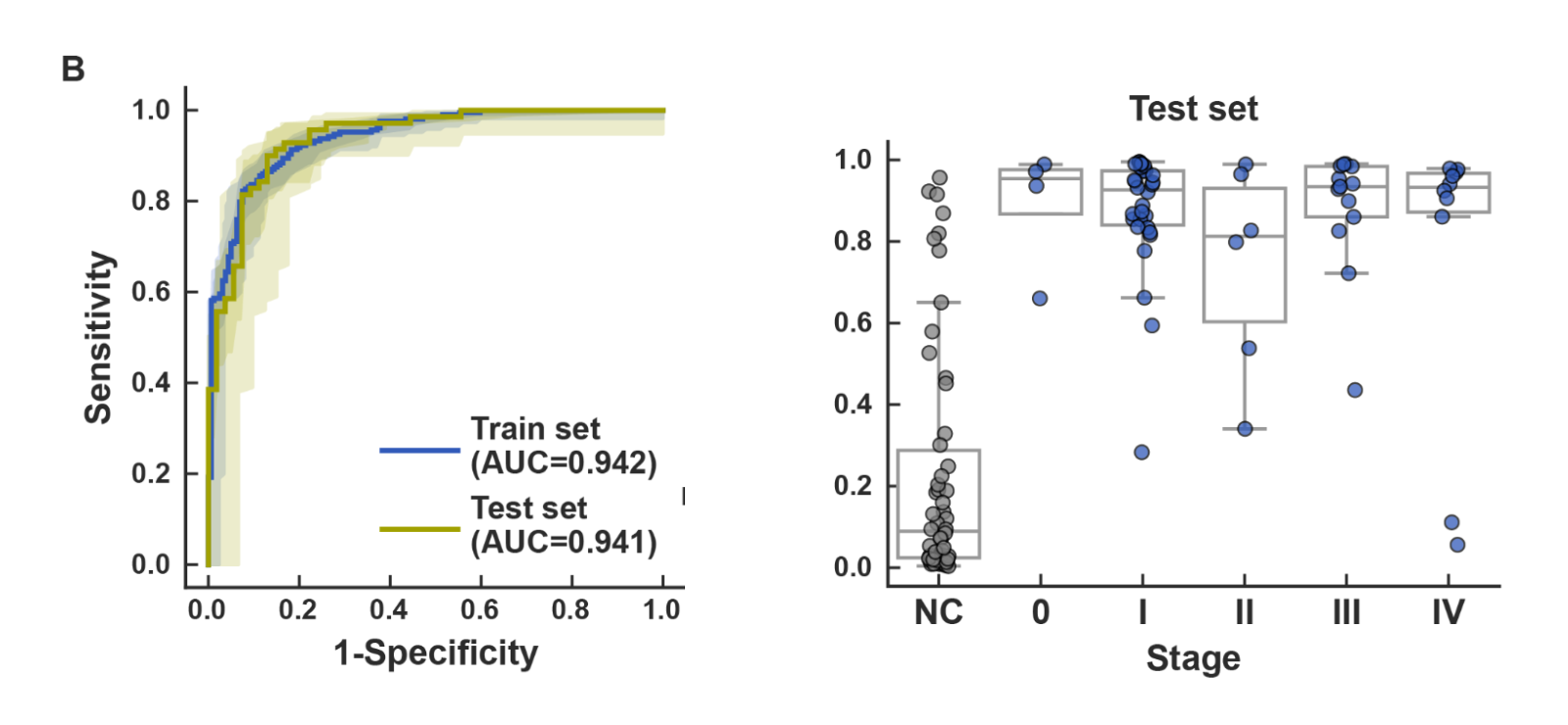
- High-accuracy early-stage lung cancer detection using urinary microRNA: By enriching and analyzing extracellular vesicle–derived microRNAs from urine and developing a machine learning model, the model achieved high diagnostic performance for early-stage lung cancer detection (test set: AUC 0.941, early-stage sensitivity 88.2%, specificity 87.0%).
- Potential for recurrence monitoring via pre- and postoperative urinary microRNA changes: Twelve microRNAs were identified that decreased following surgery and re-elevated at the time of recurrence.
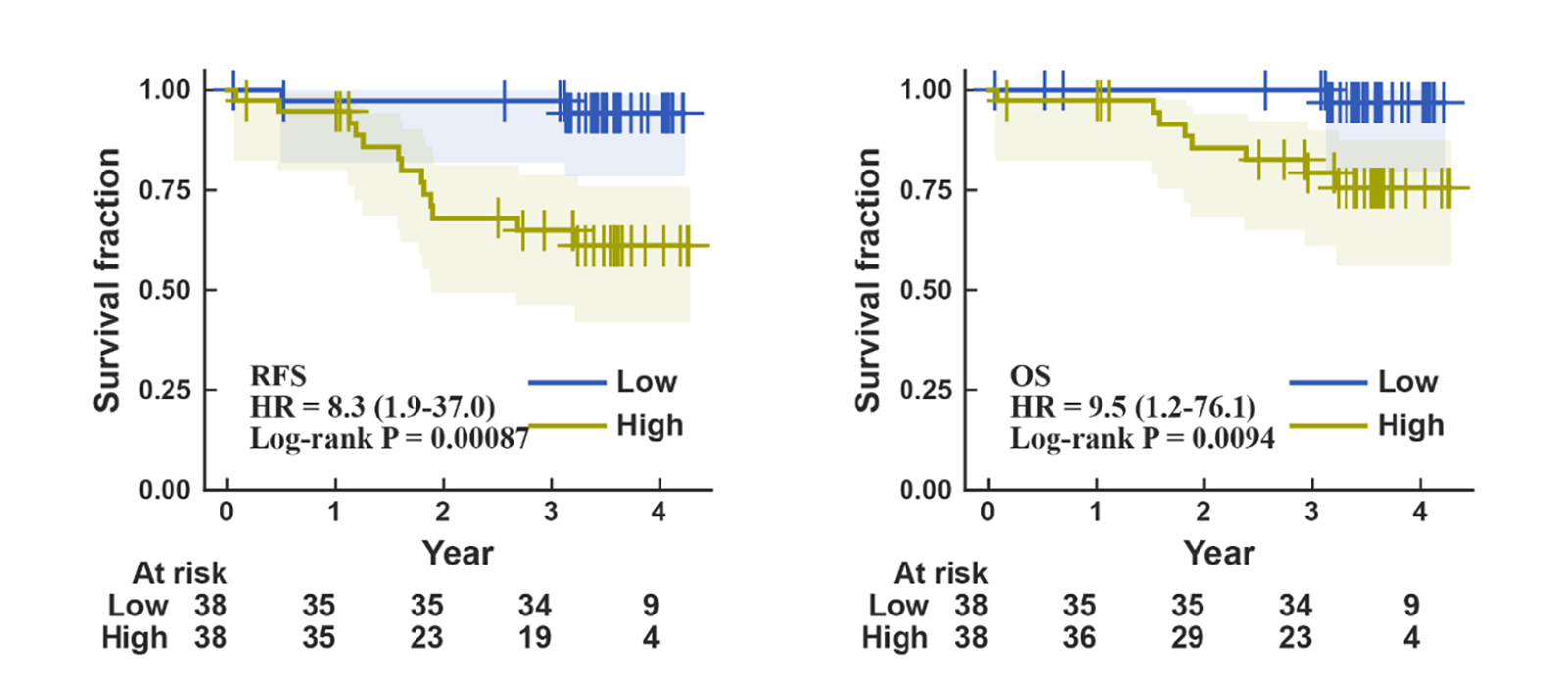
- Three-microRNA panel stratifies postoperative recurrence risk: A panel of hsa-miR-181a-5p, hsa-miR-185-5p, and hsa-miR-934 demonstrated a statistically significant difference in recurrence-free survival between high- and low-risk groups (HR = 8.3).
- Single urinary microRNA assay platform has the potential to address multiple phases of lung cancer care: The platform demonstrated the potential to comprehensively address early detection, prognosis prediction, and recurrence monitoring multiple distinct phases of lung cancer management within a single testing framework.
- Noninvasive test compatible with at-home sample collection: As the test uses urine, no blood draw is required, and samples can be collected at home. This may enable broad population-based screening and holds promise for early detection and management in regions with limited access to medical facilities.
Lung cancer is the most commonly diagnosed cancer in men and the second most common in women, and remains the leading cause of cancer-related mortality worldwide. Because symptoms are rarely present in early stages and patients are often diagnosed at an advanced stage, early detection is critical for improving clinical outcomes. Even patients who undergo surgery at an early stage carry a residual risk of postoperative recurrence. Existing blood-based tumor markers such as CEA and CYFRA21-1 have low sensitivity for early-stage lung cancer,highlighting the urgent need for more accurate, noninvasive diagnostic approaches.
The research team focused on extracellular vesicles (EVs) and microRNAs, which are closely associated with cancer cell activity. Cells communicate with distant cells via microRNAs packaged within EVs, a mechanism that cancer cells are thought to actively exploit. By concentrating urinary EVs, large quantities of microRNA can be efficiently extracted and analyzed,providing the foundation for a noninvasive urine-based lung cancer test.
Urine samples were collected from 278 lung cancer patients,approximately half of whom had early-stage disease (Stage 0/I),and 213 non-cancer controls across four institutions (The Jikei University Hospital, Higashiosaka City Medical Center, Hokuto Hospital, and Omiya City Clinic). Samples were analyzed using small RNA sequencing and machine learning.The lung cancer detection model achieved an AUC of 0.942 in the training set and 0.941 in the independent validation set. For early-stage lung cancer specifically, the model achieved sensitivity of 88.2% and specificity of 87.0% in the test set (Figure 2). Prediction scores were not significantly influenced by demographic or clinical covariates, including age, sex, BMI, and smoking history.
For recurrence monitoring applications, a comparative analysis of pre- and postoperative urine samples identified 12 microRNAs that were highly expressed in the lung cancer group, decreased following surgery, and trended toward re-elevation at the time of recurrence,suggesting their potential as biomarkers reflecting tumor presence.
To predict postoperative recurrence risk, Cox regression analysis was performed in 76 Stage I/II lung cancer patients. 11 microRNAs were significantly associated with recurrence-free survival, from which a three-miRNA panel (hsa-miR-181a-5p, hsa-miR-185-5p, hsa-miR-934) was derived. Stratification of patients into high- and low-risk groups based on this panel revealed statistically significant differences in both recurrence-free survival (HR = 8.3, p = 0.00087) and overall survival (HR = 9.5, p = 0.0094) (Figure 3).
The urinary microRNA assay platform described in this study has the potential to address multiple phases of lung cancer care, including (1) early detection, (2) prognostic stratification, and (3) recurrence monitoring, within a single integrated testing framework. As the test uses urine, it is noninvasive with minimal patient burden and compatible with at-home sample collection, raising expectations for its contribution to broad population-based screening and early detection and management of lung cancer in regions with limited healthcare access.
Journal: npj Precision Oncology
Title: A Noninvasive Urinary MicroRNA-Based Assay for Early Detection of Lung Cancer and Its Potential Application to Prognosis and Recurrence Monitoring: a case–control study
Authors: Ayu Kiritani, Shohei Mori, Takashi Nojiri, Junko Watanabe, Hiroki Yamaguchi, Yoriko Ando, Yumi Nishiyama, Mika Mizunuma, Yuki Ichikawa, Yasutaka Kato, Jun Araya, Takashi Ohtsuka, Masahiko Higashiyama, Yu Fujita
Affiliations:
1. Division of Respiratory Diseases, Department of Internal Medicine, The Jikei University School of Medicine, Tokyo, Japan
2. Division of Thoracic Surgery, Department of Surgery, The Jikei University School of Medicine, Tokyo, Japan
3. Division of Thoracic Surgery, Higashiosaka City Medical Center, Osaka, Japan
4. Craif Inc., Nagoya, Japan
5. Institute of Innovation for Future Society, Nagoya University, Nagoya, Japan
6. Tenshin Inc., Tokyo, Japan
7. Center for Cancer Genomics, Keio University School of Medicine, Tokyo, Japan
8. Department of Pathology and Genetics, Laboratory of Cancer Medical Science, Hokuto Hospital, Obihiro, Japan
9. Division of Next-Generation Drug Development, Research Center for Medical Sciences, The Jikei University School of Medicine, Tokyo, Japan
10. Center for Exosome Medical Research, The Jikei University School of Medicine, Tokyo, Japan
